Newborn screening
Introduction
Babies born with adrenoleukodystrophy (ALD) are neurologically normal at birth. However, early diagnosis of boys with ALD can lead to life-saving interventions. These include the timely initiation of adrenal steroid replacement therapy after identification of adrenal insufficiency and the provision of allogeneic hematopoietic stem cell transplantation (HSCT) as a means of treating cerebral ALD. HSCT can halt the often fatal progression of cerebral demyelination, provided the procedure is performed at a very early stage of the disease. Unfortunately, this can only be effective during a narrow therapeutic window that is often missed. Newborn screening provides access to this “window of opportunity” and allows timely initiation of these established therapies.
In February 2016, ALD was added to the US Recommended Uniform Screening Panel (RUSP). This is the federal list of all genetic conditions recommended for state newborn screening programs. New York State initiated newborn screening for ALD on December 30, 2013. Since then, the majority of U.S. states have initiated newborn screening for ALD (Fig 1). As of 2024, ALD screening is available in most states, with additional states continuing to implement programs.
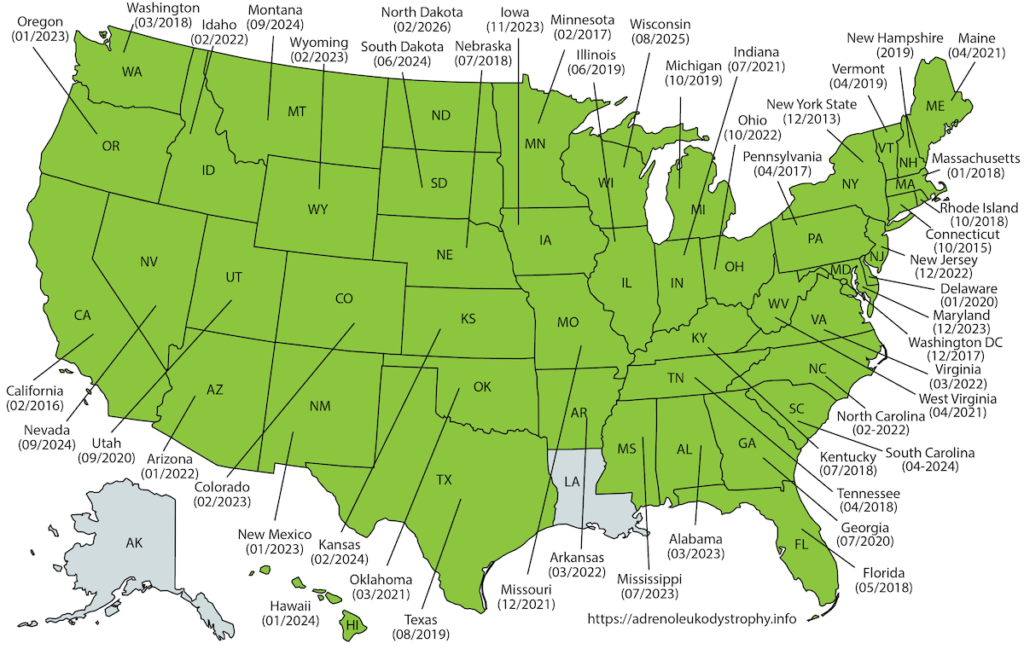
Figure 1: Map showing the states in the US that have initiated ALD newborn screening.
Outside the United States, Taiwan initiated newborn screening for ALD in November 2016 (Chen et al. 2022). Pilot programs and implementation efforts are underway in Japan (Shimozawa et al. 2021), Israel, Italy (Bonaventura et al. 2023) and Spain. Following a successful pilot study in 2021 (Albersen et al. 2022), the Netherlands initiated nationwide newborn screening for ALD on October 1, 2023. The Netherlands is the first country to implement sex-specific screening, screening only males who are at high risk for adrenal insufficiency and cerebral ALD during childhood. Boys with ALD are identified through a combination of elevated C26:0-LPC levels, sex determination (the X-counter), and confirmation of a pathogenic ABCD1 variant (Barendsen et al. 2020). Several other countries are evaluating ALD for inclusion in their screening programs.
Criteria for inclusion in the screening program
There is broad international consensus on the criteria for inclusion of a condition in a newborn screening program.
- Early diagnosis must be of direct benefit to the newborn. There must be a substantial health benefit to be gained from early intervention in serious diseases with a known natural history.
- The screening test must be of good quality. The test must have high specificity and sensitivity, i.e. a very low rate of false positive and false negative results.
 History
History
In 2004, at a meeting of the National Advisory Committee for Newborn Screening, Dr. Hugo Moser suggested that ALD be added to the U.S. RUSP. The only problem was that there was no valid test for newborn screening. To overcome this, he raised funds and recruited a team of researchers at the Kennedy Krieger Institute (Baltimore, MD) to identify a suitable biomarker and develop a test using tandem mass spectrometry (MS/MS). In 2006, the team reported the identification of C26:0-lysophosphatidylcholine (C26:0-LPC) in postnatal venous dried blood spots (DBS) from ALD males (Hubbard et al. 2006). In the following years, scientists continued to improve the analysis (Hubbard et al. 2009; Theda et al. 2014). Then, in collaboration with researchers at the Mayo Clinic (Rochester, Minnesota), a high-throughput method for the analysis of C26:0-LPC was developed (Haynes and De Jesús 2012; Turgeon et al. 2015). In 2013, this method was validated using 100,000 anonymous dried blood spots.
 Aidan’s Law
Aidan’s Law
In April 2012, after the death of their son, Aidan, who had cerebral ALD but was diagnosed too late, the Seeger family drafted and supported the passage of Aidan’s Law in New York State. The bill passed in February 2013 and became law in March 2013. On December 30, 2013, the New York State Newborn Screening Laboratory began testing babies for ALD.
New York State
In the first three years, New York State screened over 700,000 newborns and identified 45 babies with ALD: 22 boys and 23 girls (Moser et al. 2016). Based on these numbers, the birth incidence of ALD is 1 in 15,000. When a newborn with ALD is identified, the family is referred to a clinical geneticist for confirmation of the diagnosis, along with genetic counseling for support services and screening of other family members at risk for ALD (extended family screening).
In males, it is imperative to initiate serial monitoring with brain MRI to detect the earliest signs of onset of cerebral ALD and to initiate adrenal function testing to detect adrenal insufficiency. A comprehensive evaluation of neurological, neuropsychological, neuroradiological, and adrenal function is necessary because there is no test to predict the clinical outcome of an individual child born with an ALD pathogenic variant.
The newborn screening test
The details of the C26:0-LPC test may vary slightly from state/country to state/country. In general, the diagnosis of ALD is made using a three-step (tier) algorithm (Fig 2). The first step is a high-throughput standard MS/MS analysis of C26:0-LPC. Samples with elevated levels of C26:0-LPC are then screened in the second step using HPLC-MS/MS. This test is more sensitive, but also a little more time consuming. For those samples that still show elevated C26:0-LPC, sequencing of the ABCD1 gene is performed in the third step.
 Figure 2: The principles of ALD 3-tier screening.
Figure 2: The principles of ALD 3-tier screening.
Challenges: Screening boys and girls, or boys only?
There are significant challenges and ongoing ethical discussions in different countries regarding the implementation of newborn screening for ALD.
- A fundamental question in ALD newborn screening is whether to screen all newborns or only males. This debate centers on the first criterion for inclusion: early diagnosis must be of direct benefit to the newborn.
- In males, approximately one third will develop cerebral ALD between the ages of 3 and 18 years, and approximately 50% will develop adrenal insufficiency, both of which can be effectively treated when detected early. In adulthood, virtually all males will develop myeloneuropathy in adulthood, which is characterized by limb spasticity, gait dysfunction, and incontinence. Myeloneuropathy is treated symptomatically, and there is currently no disease-modifying therapy.
- Females with ALD have a <1% chance of developing adrenal insufficiency or cerebral ALD during childhood. So, newborn screening offers no direct health benefit for girls in terms of treatable childhood conditions. Approximately 80% of women with ALD will develop myelopathy by the age of 60, but like males with myeloneuropathy, this is not a condition that benefits from early childhood detection.
- Some countries, like the United States, Italy, Israel and Spain screen all newborns and identify both boys and girls. Others, like Japan and the Netherlands, have chosen to screen only males who are at risk for childhood-onset treatable conditions. Both approaches have merit, and the choice reflects differing interpretations of screening criteria and ethical frameworks.
Variant Classification and the Grey Zone
Newborn screening for ALD frequently identifies genetic variants of uncertain significance (VUS) in the ABCD1 gene. These variants are associated with borderline-elevated C26:0-LPC levels, creating diagnostic uncertainty for families. Traditional pathogenicity classifications do not adequately account for age-dependent disease penetrance or the variable clinical spectrum of ALD.
To address this challenge, risk-stratification frameworks have been developed that integrate biochemical data (C26:0-LPC levels) with genetic and clinical information. These frameworks classify variants into risk categories such as “no ALD,” “lower-risk for childhood disease,” and “at-risk for childhood disease.” This approach helps distinguish true positives from false positives, reduces unnecessary medical interventions, and provides families with evidence-based guidance about actual disease risk.
International collaboration through initiatives like the Grey Zone Project and the ABCD1 Variant Registry (adrenoleukodystrophy.info) continues to refine variant interpretation and improve screening accuracy worldwide.
Last modified | 2026-02-09

